Researchers
I am an organic chemist with strong interest in the fundamental nature of molecular interactions. I have extensive research experience in the United Kingdom and Australia including the design and synthesis of small organic molecules, organic macromolecules, peptides and medicinal compounds. I have a strong interest in molecular modelling and the use of computers to help solve research problems. In recent years I have moved my research towards a virtual in silico lab bench and have gained experience in molecular modelling and structure-based drug design.
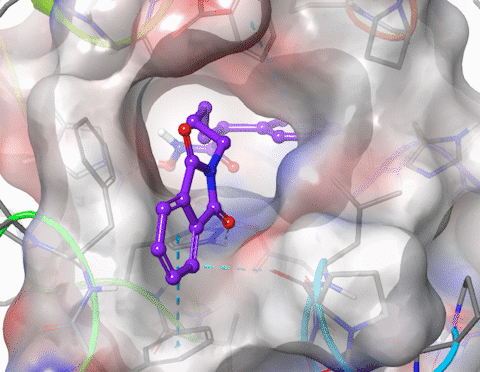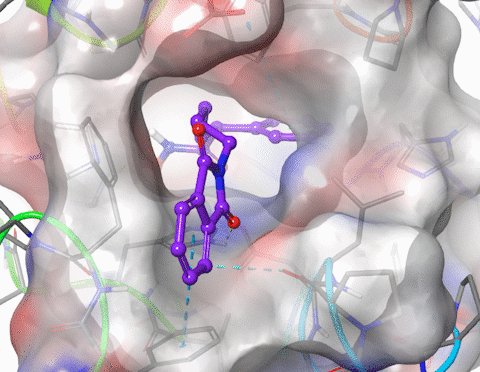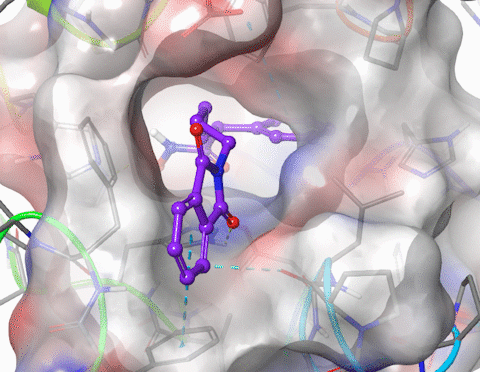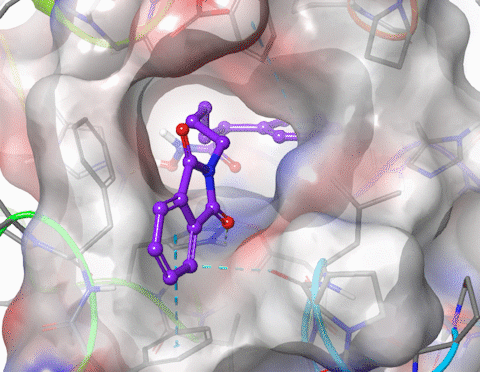
I am skilled in using molecular modelling software to develop new drug like molecules using structure-based drug design methodologies (docking and pharmacophore models). I have performed GPU-accelerated molecular dynamics to simulate large membrane spanning proteins including potassium ion channels and G-protein-coupled receptors (GPCRs). I have modelled the structure and properties of structurally constrained peptides using molecular dynamics and used cheminformatics to improve the oral bioavailability of small peptides. I have published in the fields of organic, medicinal, physical and macromolecular chemistry. Research interests include molecular modelling, medicinal chemistry (drug design, hit and lead development), organic synthesis, peptidomimetics.

Recent Publications:
1. Kim S.H.; Redvers R.P.; Chi L.H.; Ling X.; Lucke A.J.; Reid R.C.; Fairlie D.P.; Martin A.C.B.M.; Anderson R.L.; Denoyer D.; Pouliot N., Identification of brain metastasis genes and therapeutic evaluation of histone deacetylase inhibitors in a clinically relevant model of breast cancer brain metastasis. Dis Model Mech. 2018, (11), 1-16.
2. Bagnall, N. H.; Hines, B. M.; Lucke, A. J.; Gupta, P. K.; Reid, R. C.; Fairlie, D. P.; Kotze, A. C., Insecticidal activities of histone deacetylase inhibitors against a dipteran parasite of sheep, Lucilia cuprina. Int J Parasitol Drugs Drug Resist. 2017, 7 (1), 51-60.
3. Nielsen, D. S.; Shepherd, N. E.; Xu, W.; Lucke, A. J.; Stoermer, M. J.; Fairlie, D. P., Orally Absorbed Cyclic Peptides. Chem. Rev. (Washington, DC, U. S.) 2017, 117 (12), 8094-8128.
4. Harjani, J. R.; Yap, B. K.; Leung, E. W. W.; Lucke, A.; Nicholson, S. E.; Scanlon, M. J.; Chalmers, D. K.; Thompson, P. E.; Norton, R. S.; Baell, J. B., Design, Synthesis, and Characterization of Cyclic Peptidomimetics of the Inducible Nitric Oxide Synthase Binding Epitope That Disrupt the Protein-Protein Interaction Involving SPRY Domain-Containing Suppressor of Cytokine Signaling Box Protein (SPSB) 2 and Inducible Nitric Oxide Synthase. J. Med. Chem. 2016, 59 (12), 5799-5809.
5. Xu, W.; Lucke, A. J.; Fairlie, D. P., Comparing sixteen scoring functions for predicting biological activities of ligands for protein targets. J. Mol. Graphics Modell. 2015, 57, 76-88.
6. Nielsen, D. S.; Lohman, R.-J.; Hoang, H. N.; Hill, T. A.; Jones, A.; Lucke, A. J.; Fairlie, D. P., Flexibility versus Rigidity for Orally Bioavailable Cyclic Hexapeptides. ChemBioChem 2015, 16 (16), 2289-2293.
7. Perry, S. R.; Xu, W.; Wirija, A.; Lim, J.; Yau, M.-K.; Stoermer, M. J.; Lucke, A. J.; Fairlie, D. P., Three Homology Models of PAR2 Derived from Different Templates: Application to Antagonist Discovery. J. Chem. Inf. Model. 2015, 55 (6), 1181-1191.


